Oncology biosimilar case studies: rituximab biosimilars
Pharmaceutical Technology
MAY 19, 2023
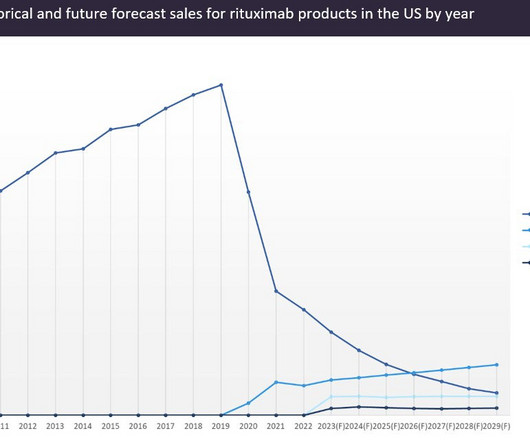
However, in the second year of biosimilar competition, Rituxan sales started to significantly erode. GlobalData forecasts Rituxan sales to plummet to just over $300m by 2029, while biosimilars will collectively surpass $1bn in sales in the same year, becoming responsible for nearly 80% of the total market share for rituximab products.











Let's personalize your content